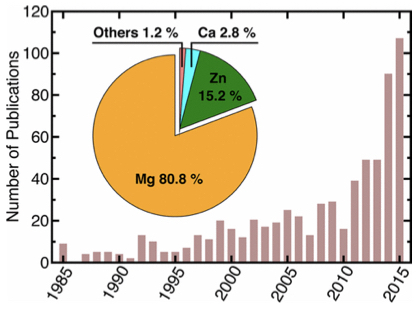
In the critical area of sustainable energy storage, solid-state batteries (see Figure 1) have attracted considerable attention due to their potential safety, energy-density and cycle-life benefits. The main proposed benefit of solid-state batteries has been their increased safety, which stems from the absence of flammable liquid electrolytes typically employed in Li-ion cells. The central electrolyte-related challenges for practical solid-state devices include utilization of metal anodes, stabilization of interfaces and the maintenance of physical contact, the solutions to which hinge on gaining greater knowledge of the underlying properties of solid electrolyte materials.
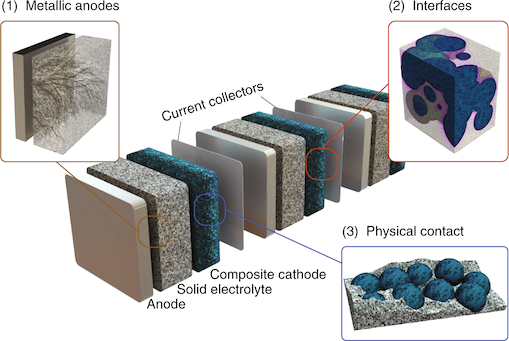
Figure 1: Representation of a bipolar-stacked solid-state battery.[1]
While significant attention is still devoted to intrinsic Li+ conductivity in solid electrolytes, many challenges remain for future solid-state applications. In the development of solid-state batteries, one the most pressing challenges are finding solid electrolytes that are electrochemically stable against electrodes, maintaining physical contact between components over many Li intercalation/extraction cycles and suppressing Li-dendrite formation.
Utilizing modeling, our research attempts to identify the root-cause of these problems, providing viable solutions.
Relevant references
- T. Famprikis, P. Canepa et al., Nature Mat. (2019)
- P. Canepa et al., Chem. Mater. 30, 9, 3019-3027 (2018)
- P. Canepa, S.-H. Bo, G. S. Gautam, B. Key, W. D. Richards, T. Shi, Y. Tian, Y. Wang, J. Li, G. Ceder.; Nat. Commun. 8, 1759 (2017)
- J. A. Dawson, P. Canepa, et al. J. Am. Chem. Soc. 140, 362–368 (2018)