Abstract
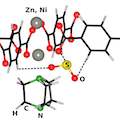
The selective adsorption of a corrosive gas, SO2, into two microporous pillared paddle-wheel frameworks M(bdc)(ted)0.5 [M = Ni, Zn; bdc =1,4-benzenedicarboxylate; ted = triethylenediamine] is studied by volumetric adsorption measurements and a combination of in situ infrared spectroscopy and ab initio density functional theory (DFT) calculations. The uptake of SO2 in M(bdc)(ted)0.5 at room temperature is quite significant, 9.97 mol/kg at 1.13 bar. The major adsorbed SO2 molecules contributing to the isotherm measurements are characterized by stretching bands at 1326 and 1144 cm–1. Theoretical calculations including van der Waals interactions (based on vdW-DF) suggest that two adsorption configurations are possible for these SO2 molecules. One geometry involves an SO2 molecule bonded through its sulfur atom to the oxygen atom of the paddle-wheel building unit and its two oxygen atoms to the C–H groups of the organic linkers by formation of hydrogen bonds. Such a configuration results in a distortion of the benzene rings, which is consistent with the experimentally observed shift of the ring deformation mode. In the other geometry, SO2 establishes hydrogen bonding with −CH2 group of the ted linker through its two oxygen atoms simultaneously. The vdW-DF-simulated frequency shifts of the SO2 stretching bands in these two configurations are similar and in good agreement with spectroscopically measured values of physisorbed SO2. In addition, the IR spectra reveal the presence of another minor species, characterized by stretching modes at 1242 and 1105 cm–1 and causing significant perturbations of MOFs vibrational modes (CHx and carboxylate groups). This species is more strongly bound, requiring a higher temperature (∼150 °C) to remove it than for the main physisorbed species. The adsorption configurations of SO2 into M(bdc)(ted)0.5 derived by infrared spectroscopy and vdW-DF calculations provide the initial understanding to develop microporous metal organic frameworks materials based on paddlewheel secondary-building units for SO2 removal in industrial processes.