Abstract
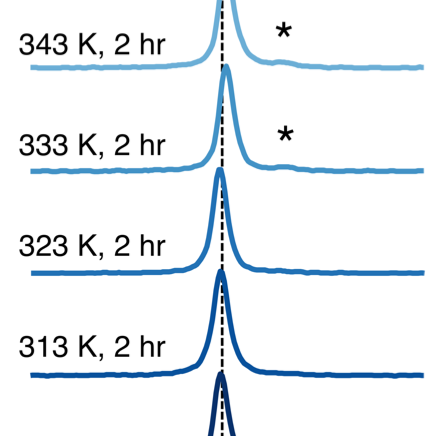
In the pursuit of urgently-needed, energy dense solid-state batteries for electric vehicle and portable electronics applications, halide solid electrolytes offer a promising path forward with exceptional compatibility against high-voltage oxide electrodes, tunable ionic conductivities, and facile processing. For this family of compounds, synthesis protocols strongly affect cation site disorder and modulate Li+ mobility. In this work, we reveal the presence of a high concentration of stacking faults in the superionic conductor Li3YCl6 and demonstrate a method of controlling its Li+ conductivity by tuning the defect concentration with synthesis and heat treatments at select temperatures. Leveraging complementary insights from variable temperature synchrotron X-ray diffraction, neutron diffraction, cryogenic transmission electron microscopy, solid-state nuclear magnetic resonance, density functional theory, and electrochemical impedance spectroscopy, we identify the nature of planar defects and the role of non-stoichiometry in lowering Li+ migration barriers and increasing Li site connectivity in mechanochemically-synthesized Li3YCl6 . We harness paramagnetic relaxation enhancement to enable 89Y solid-state NMR, and directly contrast the Y cation site disorder resulting from different preparation methods, demonstrating a potent tool for other researchers studying Y-containing compositions. With heat treatments at temperatures as low as 333 K (60°C), we decrease the concentration of planar defects, demonstrating a simple method for tuning the Li+ conductivity. Findings from this work are expected to be generalizable to other halide solid electrolyte candidates and provide an improved understanding of defect-enabled Li+ conduction in this class of Li-ion conductors.